How AI drug discovery revives productivity and slashes costs by 56%
Breaking through bottlenecks to reshape the future of pharmaceutical R&D
This is part two of a two-part exploration of pharmaceutical productivity decline, how process analysis can clarify its root causes, and which levers AI drug discovery might pull to reduce the costs to bring a drug to market. Click the following link to read part one:
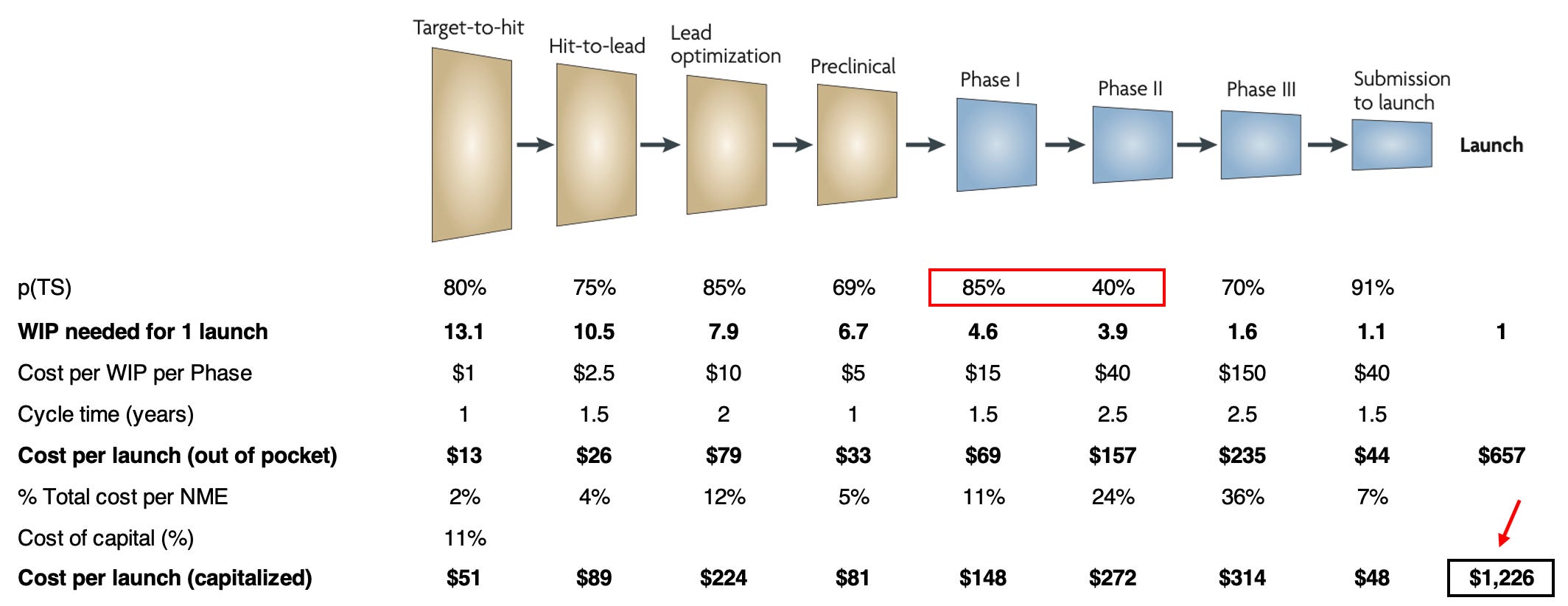
In part one, we looked at the decline of productivity in the pharmaceutical industry. Decreasing sales and rising costs have pushed the average pharmaceutical internal rate of return (IRR) toward 0%. A widely cited process model by Steven Paul shows how adjustments to the cost, duration, and risk of each stage in drug development affects the capitalized cost — the time-value adjusted total cost to bring one drug to market. This metric serves as our “north star”. Lower capitalized cost improves IRR and makes drug development financially viable.
Paul’s drug process model, published in 2010, estimated the average capitalized cost of a drug at $1.78B. Today, it may exceed $6B. While the absolute numbers are staggering, what matters most is the percentage change that AI can deliver. We’ll base all our estimates on the assumptions of 2010. Again, for an explanation of the model, refer to part one.
The application of AI to drug discovery is heralded as a remedy for the industry’s productivity woes. Many claims about AI’s potential to reduce costs and timelines are backed by quantitative estimates. Conveniently, we can plug them directly into our model to see the percentage decrease in capitalized cost.
1. Faster and cheaper discovery — 25% cost reduction
If you read my post about AI in education versus the workplace, you’ll know that AI excels at ideation, particularly evident when applied in a materials engineering company.
The pharmaceutical industry faces a similar situation. Just as AI can rapidly generate new material compositions, it can also automate the creation of novel molecular arrangements during the discovery phases of drug development. In essence, the discovery activities in pharma, akin to ideation in materials engineering, are poised to gain the most immediate advantage from AI-driven innovation.
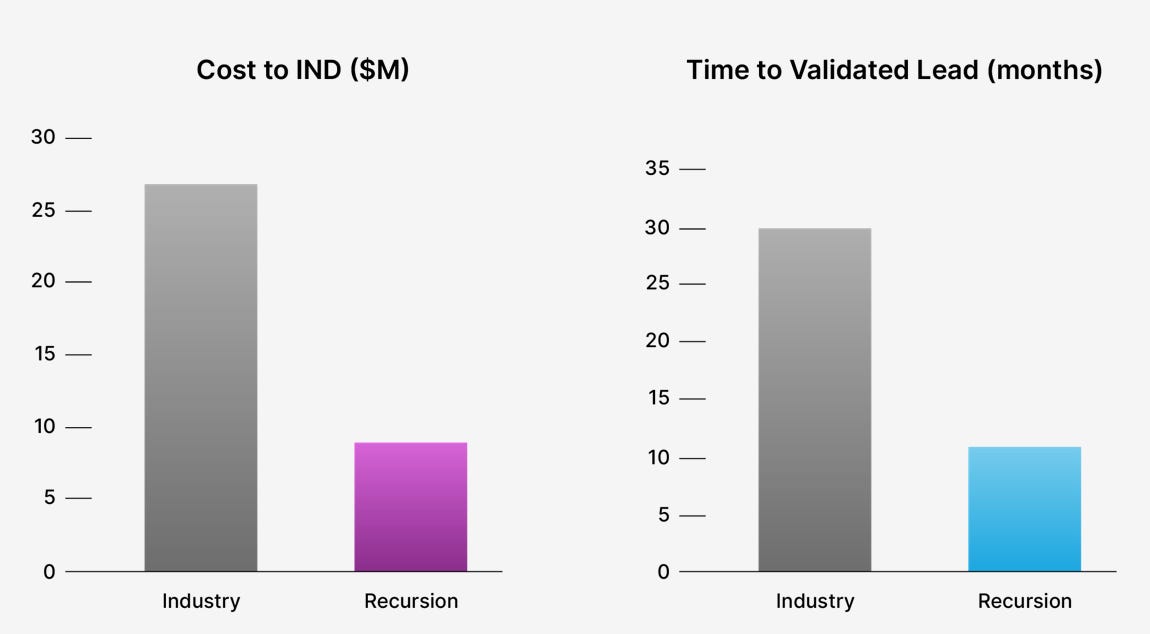
Recursion Pharmaceuticals, an AI-driven drug discovery company founded in 2013, exemplifies this approach. The company has formed partnerships with industry leaders like Roche, Sanofi, and Bayer. Recursion claims significant reductions in both “cost to IND” and “time to validated lead,” analogous to the hit-to-lead and lead optimization phases in Paul’s model. These steps together take a large number of molecules that bind or affect the intended biological target and arrive at a few optimized candidates.
So we’ll represent this as a 70% reduction in cost and set the duration to 6 months for both phases:
The new capitalized cost of $1.3B represents a 25% drop. Most of this is due to lead optimization being a relatively expensive process ($10M without AI) that you need to do with a lot of candidates (14.6). Lowering the cycle time also “pulls forward” the subsequent phases on the timeline, reducing the impact of time value of money.
Notably, this says nothing about how likely these candidates are to pass clinical trials. We’re making the assumption that the AI is equally as skilled at selecting and optimizing compounds as a human would be, just cheaper and faster.
But it’s not inconceivable that an AI powered by a battalion of Nvidia GPUs could “see ahead” and prioritize candidates with better downstream safety and efficacy profiles. So what does the data say?
2. Improved Phase 1 and 2 conversion — 31% cost reduction
Clinical trials are the bottleneck of drug development. They take a long time and cost tens of millions of dollars. Any improvement in success rate is therefore highly impactful. The data is somewhat scant, but it suggests AI-discovered candidates are more likely to pass Phase 1 (safety) and potentially Phase 2 (efficacy).
A Nature paper looked at 75 AI-discovered drug candidates in clinical trials today. 80-90% passed Phase 1. That’s a remarkable jump from the 54% achieved by humans.
It may be that the AI only makes fine-tuning adjustments to well-established molecular classes. In other words, the AI isn’t rocking the boat too much, so there’s a minimal chance that new candidates will produce toxic effects. Or, it could just be that that AI is better than humans at identifying a drug’s safety characteristics. The fact that all molecular classes in the study (antibodies, vaccines, small molecules, repurposed drugs) experienced the same Phase 1 benefit points to this being at least partially true.
Phase 2 was ambiguous. 40% of drugs succeeded on efficacy, albeit with a small sample size. With that said, four of the six of these drugs discontinued after Phase 2 were for business reasons rather than clinical challenges. So I’m inclined to give the benefit of the doubt.
Given capitalized cost is highly sensitive to clinical trial success probability, it’s not surprising that the impact is significant. Clinical trials are massively expensive, and according to our model we need 8.6 Phase 1 and 4.6 Phase 2 trials just to launch one drug. With our improved odds of success with AI, we now only need 4.6 Phase 1 and 3.9 Phase 2 trials.
At the top of the funnel, only 13.1 candidates need to be introduced to yield one drug, versus 24.3 initially. The odds of any single candidate passing the entire process are about twice as high with AI.
3. Clinical trial optimization — 19% cost reduction
Beyond candidate selection, AI can streamline clinical trials in other ways. A McKinsey report highlights AI’s ability to:
Optimize patient recruitment, reducing trial sizes.
Enhance genotypic and phenotypic understanding of patient responses.
Analyze imaging data (MRI, CAT scans, X-rays) to evaluate drug impacts.
McKinsey gives these improvements credit for a 20% reduction in cost and duration.
Again, since clinical trial costs are such a huge lever, it’s not surprising that a 20% reduction translates to dropping cost per launch by an equal amount.
4. All improvements combined — 56% cost reduction
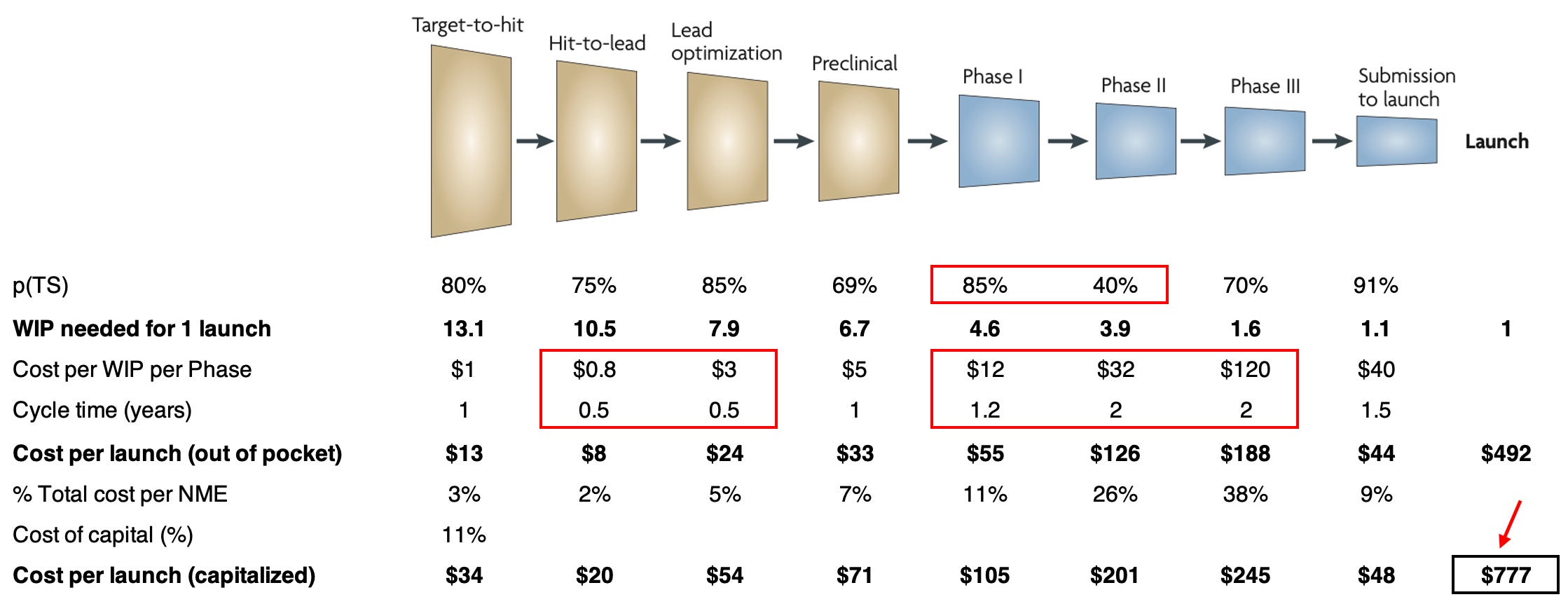
What would a drug discovery process look like if a drug were brought to market using faster and cheaper discovery, improved clinical trial success rates, and streamlined trial execution?
When we combine all these factors, the cumulative impact is striking. The capitalized cost, originally modeled at $1.8B, now drops to about $777M. That’s more than a 50% reduction, a remarkable move toward restoring the pharmaceutical IRRs of decades past.
Beyond the cost savings, this enhanced process also reduces the total time it takes for a drug to move from initial conception to market launch. Fewer compounds are needed at the start of the pipeline, and improved efficiencies at every stage mean less waiting, fewer failures, and more predictable outcomes. Patients will benefit from quicker access to innovative treatments, while companies enjoy more sustainable and attractive returns on their R&D investments.
Final notes
Of course, all of this is still a bit uncertain. Phase 2 data remains limited, and it’s possible that the AI advantage diminishes once it’s picked off the easiest targets. Maybe at some point we’ll hit a “quality cliff,” where the AI’s ability to find new high-quality candidates tapers off.
But I’m inclined to stay optimistic. Clinical trial success rates are the biggest lever in controlling capitalized cost, and AI seems to have an empirically-supported impact.
Even if part of the initial Phase 1 success is due to AI’s focus on safer, well-understood chemical families, there’s evidence that AI has also ventured into unknown spaces. That suggests we’re not just making the old stuff cheaper and faster; we’re building a new pipeline that could ultimately reshape what’s possible in medicine. From the Nature paper:
Earlier analysis suggests that AI algorithms have been used to explore novel chemical space and not just to fine-tune previously known structures.
While there’s still a lot we don’t know, this early data makes it hard not to feel bullish. AI might just boost the industry enough to reclaim its lost productivity and bring more innovative treatments to patients sooner.







Great read Mark! Well researched. Insightful and thought provoking.